
 Partnership
Partnership
EMBL and Institut Pasteur: a flagship European partnership to support interdisciplinary scientific collaboration in infection biology
Since strengthening their links at the "Building bridges in infection biology" workshop in July 2024 (see the newsletter from June 28, 2024), the European Molecular Biology Laboratory (EMBL) and the Institut Pasteur have embarked on a partnership strategy to step up their collaboration in the field of infectious disease biology.
They will draw on their shared expertise to explore essential topics such as pathogen mechanisms, host-pathogen interactions, the influence of human genetics on susceptibility to infection and innovative strategies to tackle antimicrobial resistance, making use of deep learning, computational biology, imaging and structural biology techniques to drive this unprecedented partnership between two leading European institutions.
Collaborative projects are already receiving valuable support from EMBL's "Amplifying Funds in Infection Biology" initiative. An incentive scheme has also been set up to provide funding for early career scientists at the Institut Pasteur to travel or complete training within the EMBL ecosystem.
 Initiative Amplifying Funds in Infection Biology
Initiative Amplifying Funds in Infection Biology
The Institut Pasteur has joined EMBL's Amplifying Funds in Infection Biology initiative, aimed at encouraging interdisciplinary research in infection biology across Europe via collaborative research projects.
This funding supports joint projects in all fields of infection biology between group leaders at the Institut Pasteur and collaborating institutions: EMBL (all European sites), the Laboratory for Molecular Infection Medicine Sweden (MIMS), the Helmholtz Centre for Infection Research (HZI) in Germany and the Wellcome Sanger Institute in the UK.
A joint call for proposals was launched by EMBL and the Institut Pasteur in September 2024 to select research projects exploring essential aspects of infection biology such as pathogen-specific protein machineries, host-pathogen interactions at different scales, how human genetics influences susceptibility to infection, and innovative approaches to tackling antimicrobial resistance. The call was also open to proposals on the human microbiome or other microbiomes related to infection.
Of the 17 proposals submitted, seven were selected for funding, five of which involve the Institut Pasteur. Each of the five funded projects will receive €50,000 per institute per year over two years:
![]() Towards a mechanistic understanding of NK cell memoryco-supervised by Mélanie Hamon, head of the Chromatin and Infection unit.
Towards a mechanistic understanding of NK cell memoryco-supervised by Mélanie Hamon, head of the Chromatin and Infection unit.
 This study aims to understand natural killer (NK) cell memory in bacterial infections, using as a model Streptococcus pneumoniae, the leading cause of bacterial pneumonia worldwide. Traditionally associated with innate immunity, NK cells have been shown to develop memory-like properties which could be harnessed for new immunotherapy approaches against bacterial pathogens. The study’s goals are to identify specific marker genes for memory NK cells and to investigate key chromatin regulators essential for NK cell memory maintenance, ultimately contributing to an unprecedented understanding of this process during bacterial infection.
This study aims to understand natural killer (NK) cell memory in bacterial infections, using as a model Streptococcus pneumoniae, the leading cause of bacterial pneumonia worldwide. Traditionally associated with innate immunity, NK cells have been shown to develop memory-like properties which could be harnessed for new immunotherapy approaches against bacterial pathogens. The study’s goals are to identify specific marker genes for memory NK cells and to investigate key chromatin regulators essential for NK cell memory maintenance, ultimately contributing to an unprecedented understanding of this process during bacterial infection.
![]() Exploring the role of translation landscapes in Salmonella pathogenicity and antibiotic resistance during the colonization of diverse host intracellular niches co-supervised by Jost Enninga, head of the Dynamics of Host-Pathogen Interactions unit.
Exploring the role of translation landscapes in Salmonella pathogenicity and antibiotic resistance during the colonization of diverse host intracellular niches co-supervised by Jost Enninga, head of the Dynamics of Host-Pathogen Interactions unit.
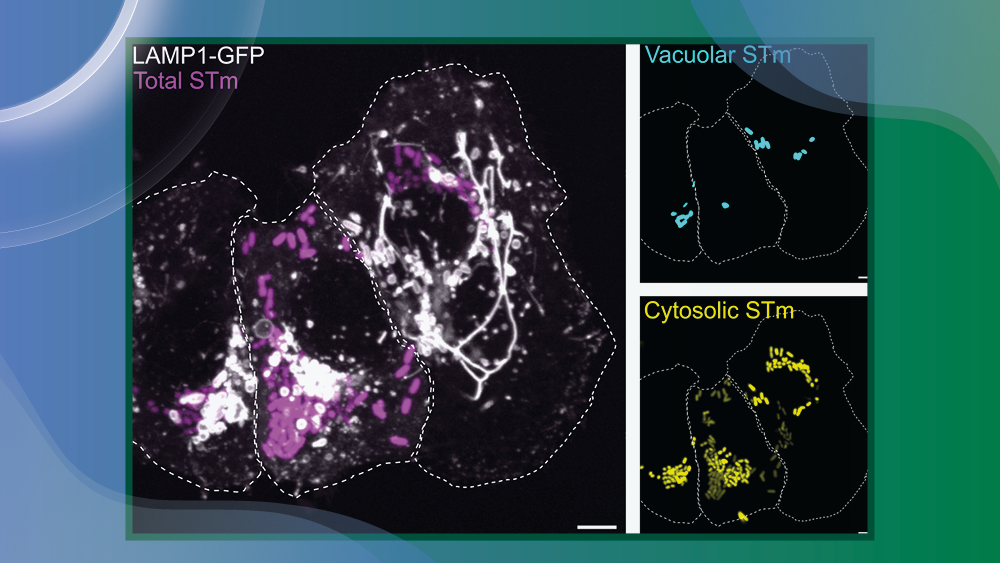 This project aims to elucidate the intracellular Salmonella Typhimurium (S.Tm) translation landscapes to better understand differences related to the colonisation of diverse host-cell niches. We will employ cryo-correlative light and electron microscopy alongside subtomogram averaging to structurally analyze S.Tm ribosomes in distinct growth states of the pathogen that are related to specific intracellular localization within infected cells. This innovative approach will allow us to simultaneously pinpoint bacterial localization and ribosomal conformations, shedding light on the pathogen’s adaptation strategies. Finally, we will assess the impact of ribosome-targeting antibiotics on the translation machinery across the different niches. This study will deepen our understanding of intracellular bacterial pathogenesis, identify factors contributing to antibiotic resistance, and explore novel therapeutic strategies.
This project aims to elucidate the intracellular Salmonella Typhimurium (S.Tm) translation landscapes to better understand differences related to the colonisation of diverse host-cell niches. We will employ cryo-correlative light and electron microscopy alongside subtomogram averaging to structurally analyze S.Tm ribosomes in distinct growth states of the pathogen that are related to specific intracellular localization within infected cells. This innovative approach will allow us to simultaneously pinpoint bacterial localization and ribosomal conformations, shedding light on the pathogen’s adaptation strategies. Finally, we will assess the impact of ribosome-targeting antibiotics on the translation machinery across the different niches. This study will deepen our understanding of intracellular bacterial pathogenesis, identify factors contributing to antibiotic resistance, and explore novel therapeutic strategies.
![]() Bioengineering vascularized human microlivers for malaria infection co-supervised by Liliana Mancio-Silva, head of the Plasmodium Infection and Transmissiongroup.
Bioengineering vascularized human microlivers for malaria infection co-supervised by Liliana Mancio-Silva, head of the Plasmodium Infection and Transmissiongroup.
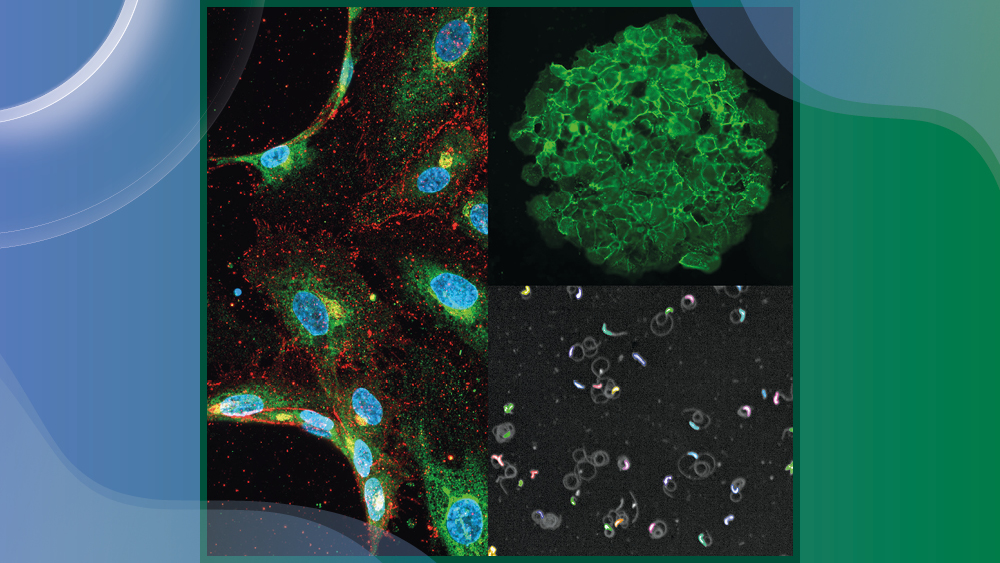 This project will bioengineer new in vitro platforms that capture the complexity of the human liver to investigate host-parasite interactions with the liver microvasculature and their impact on hepatocyte infection. Specifically, we aim to characterise parasite interactions with engineered human 3D liver microvessels and to build a vascularised human microliver to shed light on receptor-ligand interactions driving early infection. This project will lay the foundation for a deeper understanding of potentially targetable host-pathogen interactions and generate a preclinical platform aimed at evaluating therapeutic interventions.
This project will bioengineer new in vitro platforms that capture the complexity of the human liver to investigate host-parasite interactions with the liver microvasculature and their impact on hepatocyte infection. Specifically, we aim to characterise parasite interactions with engineered human 3D liver microvessels and to build a vascularised human microliver to shed light on receptor-ligand interactions driving early infection. This project will lay the foundation for a deeper understanding of potentially targetable host-pathogen interactions and generate a preclinical platform aimed at evaluating therapeutic interventions.
![]() Exploring new therapeutic opportunities against mpox virus co-supervised by Pablo Guardado-Calvo, head of the Structural biology of infectious diseases five-year group.
Exploring new therapeutic opportunities against mpox virus co-supervised by Pablo Guardado-Calvo, head of the Structural biology of infectious diseases five-year group.
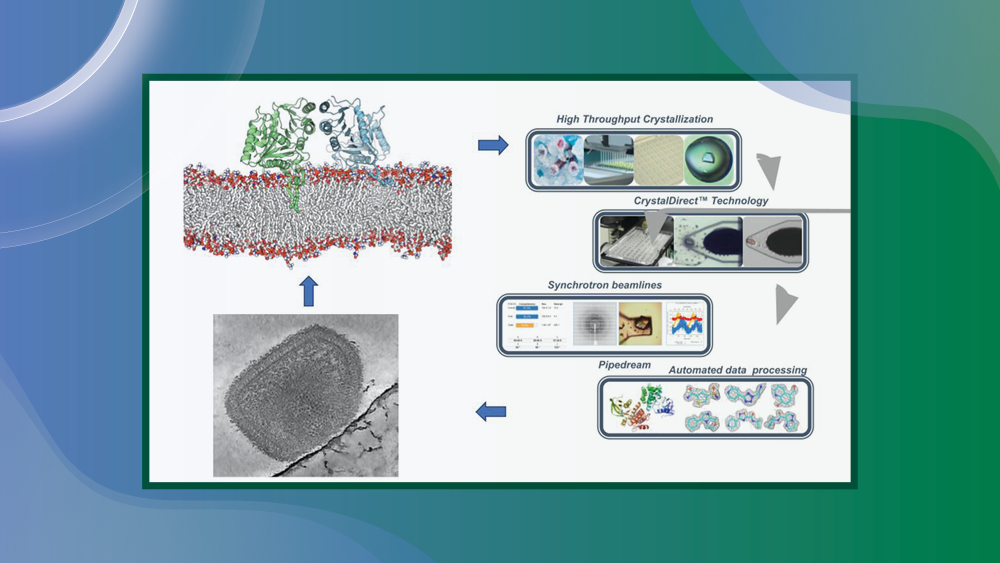
Today, there are concerns that smallpox re-emerge as a bioweapon or the emergence of zoonotic orthopoxviruses, as seen with the mpox outbreaks in 2022 and 2024. Current treatments for poxvirus infections are effective but limited: first-generation vaccines are discontinued and modern vaccines are challenging to produce on a large scale and fail to produce long-term immunity. Tecovirimat, the primary treatment for mpox, is vulnerable to viral resistance as a single mutation in the viral phospholipase F13 may confer resistance to the drug. There is a pressing need for developing broad-spectrum antivirals to treat mpox and other diseases caused by orthopoxviruses.
This project aims to combine the expertise of EMBL and the Institut Pasteur to establish proof of concept for novel therapeutic strategies that could lead to the development of new antivirals targeting different pockets of the F13 protein.
![]() Personalizing gut decolonization of MDR Enterobacteria through a systems biology approach co-supervised by David Bikard, head of the Synthetic Biology unit.
Personalizing gut decolonization of MDR Enterobacteria through a systems biology approach co-supervised by David Bikard, head of the Synthetic Biology unit.
 This project aims to identify the genetic factors in pathogens and microbiota members that determine MDR-E gut colonisation to support the development of synbiotic interventions, such as the combination of pre-/probiotic modalities, for removing MDR-E from the gut. This objective will be achieved by combining the complementary expertise of research teams in microbiome and infection biology. The long-term goal is to stratify individuals colonised with MDR-E into rationally designed synbiotic interventions based on their microbiota composition.
This project aims to identify the genetic factors in pathogens and microbiota members that determine MDR-E gut colonisation to support the development of synbiotic interventions, such as the combination of pre-/probiotic modalities, for removing MDR-E from the gut. This objective will be achieved by combining the complementary expertise of research teams in microbiome and infection biology. The long-term goal is to stratify individuals colonised with MDR-E into rationally designed synbiotic interventions based on their microbiota composition.
Find out more about the results of the Amplifying Funds in Infection Biology initiative
 Travel meeting et Training grants
Travel meeting et Training grants
As well as these collaborative projects, the strategic initiative between EMBL and the Institut Pasteur led the Department for Scientific Programming and Incentive Actions to launch the first session of the Travel and Training Grants program in autumn 2024, to fund travel and training for early career scientists on campus (see the newsletter article on October 11).
This program gives early career scientists the opportunity to attend conferences organized by EMBL or complete training at EMBL research facilities or units on its various European sites.
A total of 21 grants (ranging from €1,000 Travel Grants to €4,000 Training Grants, for a total Institut Pasteur budget of €36,000) have recently been awarded in connection with this incentive program.
A second session will be held during spring 2025.
 Key people involved in the partnership
Key people involved in the partnership
Several Institut Pasteur staff are closely involved in the partnership between EMBL and the Institut Pasteur.
Scientific staff:



Pascale Cossart, Professor at the Institut Pasteur and Honorary Permanent Secretary at the French Academy of Sciences
Sebastian Baumgarten,Head of the Parasite RNA Biology G5
Felix Rey, Head of the Structural Virology Unit
Support staff:


Mallory Perrin-Wolf, Head of the Department for Scientific Programming and Incentive Actions
Guillaume Frasca, Coordinator for Scientific Affairs